In the specialized field of chemical synthesis and polymer formulation, the stability of the solvent often dictates the safety and quality of the final product. While botanically derived terpenes such as Limonene are celebrated for their renewable origins and solvency power, their inherent instability poses significant challenges in industrial applications. Specifically, the presence of unsaturated double bonds in Limonene makes it susceptible to rapid oxidation, leading to viscosity changes and the formation of hazardous peroxides. For formulation scientists and procurement managers, the solution lies in Paramenthane—a fully hydrogenated, saturated derivative that retains the bio-based advantages of terpenes while offering the chemical inertness required for sensitive reactions and long-term storage.
The Science of Saturation: Transforming Limonene into Paramenthane
Industrial applications require solvents that remain consistent under stress. The transformation of raw terpenes into stable industrial media begins with understanding the botanical derived terpenes meaning in a chemical context. These compounds, extracted primarily from citrus peels and pine resins, serve as the feedstock. However, their raw utility is limited by their molecular structure.
From Unstable Precursors to Inert Solutions
The fundamental difference between Limonene and Paramenthane lies in their bond structures. Limonene (C10H16) contains unsaturated carbon-carbon double bonds. These sites are chemically reactive, making the molecule prone to autoxidation when exposed to air or heat. This degradation not only alters the sensory profile—a topic frequently debated in botanically derived terpenes reddit communities regarding consumer products—but more critically, it compromises industrial safety by generating unstable hydroperoxides.
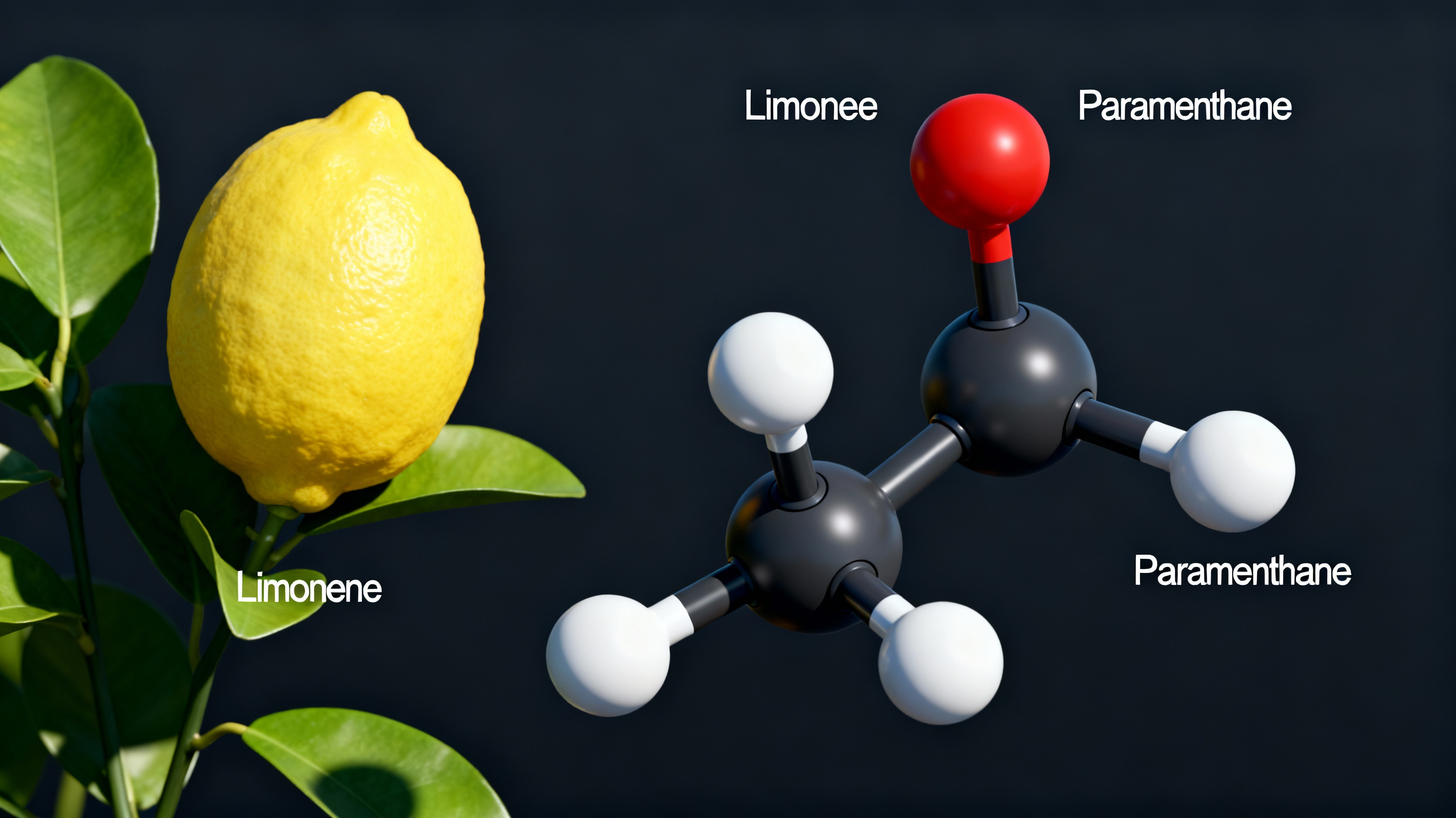
To neutralize this risk, the industry employs catalytic hydrogenation. In this process, hydrogen atoms are added to the Limonene molecule, breaking the double bonds and converting them into single, stable bonds. The result is Paramenthane (C10H20), a saturated monocyclic terpene. This saturation renders the molecule chemically inert, effectively immunizing it against oxidative aging. For chemical engineers, this means the solvent acts purely as a medium, refusing to participate in unwanted side reactions or degrade during extended storage periods.
Strategic Industrial Applications and Market Distinctions
The utility of terpenes varies drastically depending on the end-use sector. This divergence is most evident when analyzing cannabis-derived terpenes vs botanical terpenes. While the former prioritizes aromatic complexity and the “entourage effect” for consumer goods, industrial botanical terpenes like Paramenthane prioritize absolute purity and lack of reactivity.
Phlegmatization and Process Safety
The primary industrial driver for switching to Paramenthane is safety in polymer production, specifically in the phlegmatization of organic peroxides. Organic peroxides are essential initiators for polymerization but are notoriously shock-sensitive and thermally unstable. A phlegmatizing agent acts as a diluent to desensitize these compounds, allowing for safe transport and handling.
Paramenthane is superior in this role because its saturated structure prevents it from reacting with the peroxide itself. Unlike unsaturated terpenes that might accelerate decomposition or induce thermal runaway, Paramenthane maintains a neutral chemical environment. This thermal stability ensures that the peroxide remains active only when triggered in the reactor, not during logistics.
Inert Media for Precision Synthesis
Beyond safety, the inert nature of Paramenthane makes it an ideal solvent for cannabis derived terpenes vs botanical terpenes discussions regarding purity in reaction media. In pharmaceutical and agrochemical synthesis, solvents must facilitate the reaction without interfering. Paramenthane’s resistance to free radical attacks allows it to be used in radical halogenation and other sensitive processes where traditional solvents might degrade or generate impurities. Furthermore, as a bio-based alternative to chlorinated solvents and aromatic hydrocarbons like Toluene, it supports Environmental, Health, and Safety (EHS) compliance by offering improved biodegradability without sacrificing solvency power.
Linxingpinechem Paramenthane Product Overview
Linxingpinechem specializes in the deep processing of pine chemicals, leveraging advanced hydrogenation technologies to produce high-purity Paramenthane designed for critical industrial applications. By controlling the hydrogenation process with precision, Linxingpinechem ensures a solvent that meets the rigorous demands of global chemical manufacturers.
Key Specifications and Advantages
- High Purity & Consistency:The product features a purity of ≥ 95%, ensuring minimal batch-to-batch variation that could affect formulation stability.
- Superior Stability:With an Iodine Value of ≤ 0.8, the solvent indicates near-complete saturation, confirming its resistance to oxidation and suitability for long-term storage.
- Physical Properties:It presents as a colorless, transparent oily liquid with a Refractive Index between 1.437 and 1.460, making it easy to integrate into clear formulations without altering visual quality.
- Optimized Impurity Profile:The presence of p-Cymene is strictly controlled to ≤ 0.5%, reducing the risk of aromatic contamination in sensitive synthesis environments.
Industrial Suitability
Linxingpinechem Paramenthane is engineered specifically for use as a safe phlegmatizer for organic peroxides and a non-reactive solvent for hydrogen peroxide precursors. Its robust stability profile also makes it an excellent candidate for replacing petrochemical solvents in cleaning applications and specialized reaction media where oxidative resistance is paramount.
Conclusion
Transitioning from reactive Limonene to stable Paramenthane represents a critical optimization for industries reliant on bio-based solvents. By choosing a hydrogenated solution, manufacturers can secure the environmental benefits of botanical sourcing while ensuring process safety and product consistency. To access detailed technical data sheets or discuss supply continuity for high-purity Paramenthane, contact the Linxingpinechem technical team today.

